SF - Immunization Devices
Download file
Product Details
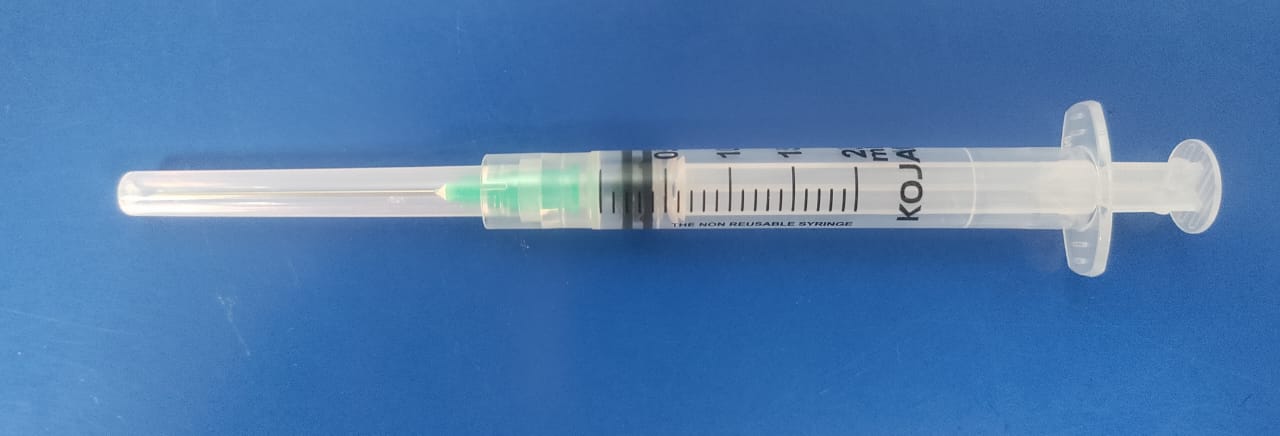
Product name
2ml KOJAK SELINGE Auto disable Syringe
IMD-PQS Code
E008-126
Status
Prequalified
Date of acceptance
24 Mar 2026
Appliance type
E008
Product description
AD syringe 2ml
Manufacturer
Hindustan Syringes & Medical Devices Ltd.
Manufacturer Reference
2ml KOJAK SELINGE Auto disable Syringe
Country of Manufacture
India
Address
174, 178/25 Ballabgarh
Faridabad
Haryana
India
Faridabad
Haryana
India
Telephone
+91 98 102 90320
Website address
Valid until
31 May 2027
Specifications
Product specifications - Main
Vaccine capacity (mL)
2.00
Graduations (ml)
0.10
Syringe material(s)
Polypropylene
Packaging & Sterility Assurance
Yes
Auto-disabling at:
End of injection
Needle Size
Other
Needle Size (Other)
21Gx1.5" (0.80x38mm)
AD Sub Category
Simple AD
AD Mechanism
Locked / Broken Plunger
AD Primary packaging
Blister & ribbon pack
Price and shipping
Incoterms
FCA
Quality standard
Quality Standard
ISO 13485;Other
Quality standard (Other)
ISO 7886-4:2018; ISO 7864:2016; ISO 23908
Specification Reference
ISO 7886-3:2020
Product Sites
Hindustan Syringes & Medical Devices Ltd.
99/25, Ballabgarh
Faridabad,
Haryana
121004
India
Document Details