WHO Product ID
AP002
Status
Prequalified
INN, dosage form and strength
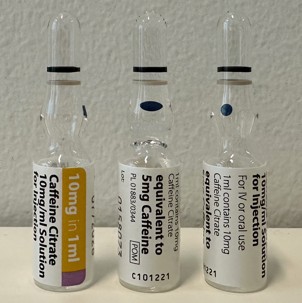
Caffeine citrate Solution for injection 10mg/ml
Date of prequalification
Basis of listing
Prequalification - Abridged
Therapeutic area
Apnoea
Type
Finished Pharmaceutical Product
Dosage form
Solution for injection
Applicant organization
Macarthys Laboratories Limited
Bampton Road, Harold Hill, Romford
Essex,
RM3 8UG
United Kingdom of Great Britain and Northern Ireland
Packaging details and storage conditions
Packaging Type
Ampoule, Type 1 glass
Configuration
1ml x 10
Shelf life (months)
36
Storage conditions
Do not store above 30°C. Do not freeze
API Manufacturing Site(s)
By Organization
By Active Ingredient
Siegfried Pharmachemikalien Minden GmbH
Karlstrasse 15-39, 42-44,
Minden,
32423
Germany
Caffeine citrate
FPP Manufacturing Site(s)
Laboratories Sterop
Scheutlaan 46-50, Anderlecht
Brussels,
B-1070
Belgium
Macarthys Laboratories Limited
Bampton Road, Harold Hill, Romford
Essex,
RM3 8UG
United Kingdom of Great Britain and Northern Ireland