WHO Product ID
RH062
Status
Prequalified
INN, dosage form and strength
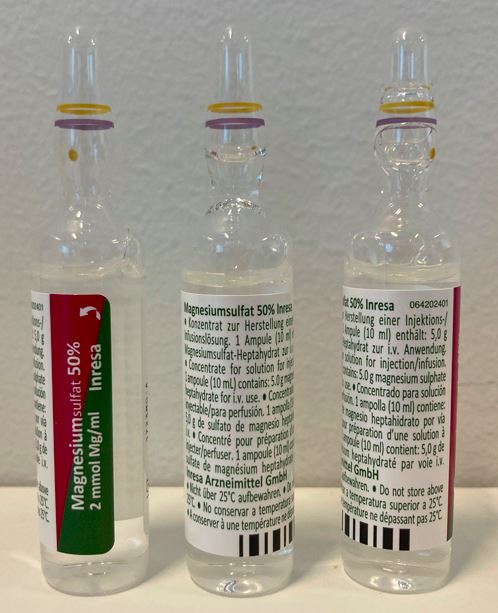
Magnesium sulphate Solution for injection 500mg/mL
Date of prequalification
Basis of listing
Prequalification - Abridged
Therapeutic area
Reproductive health
Type
Finished Pharmaceutical Product
Dosage form
Solution for injection
Applicant organization
Inresa Arzneimittel GmbH
Obere Hardtstraße 18, 79114
Freiburg,
Germany
Packaging details and storage conditions
Packaging Type
Ampoule, Type I glass
Configuration
10mlx5,10mlx10,10mlx50,10mlx100
Shelf life (months)
60
Storage conditions
Do not store above 30°C
API Manufacturing Site(s)
By Organization
By Active Ingredient
K+S KALI GmbH
Bertha-von-Suttner-Strasse 7
KASSEL,
Germany
Magnesium sulphate
Macco Organiques, S.R.O
Zahradni 1938/46c
Bruntál,
792 01
Czechia
Magnesium sulphate
FPP Manufacturing Site(s)
Laboratoire RENAUDIN
ZA Errobi, 64250, ITXASSOU
France