WHO Product ID
HA692
Status
Prequalified
INN, dosage form and strength
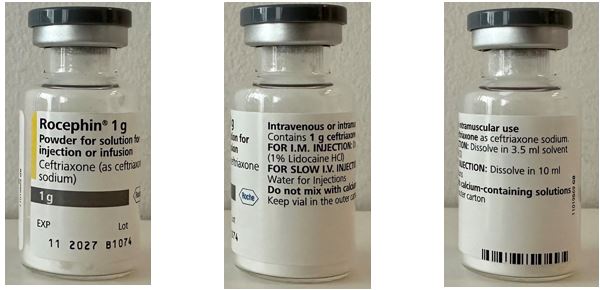
Ceftriaxone (sodium) Powder for solution for injection 1g
Date of prequalification
Basis of listing
Prequalification - Abridged
Therapeutic area
HIV/AIDS
Type
Finished Pharmaceutical Product
Dosage form
Powder for solution for injection
Applicant organization
Roche Products Limited
Hexagon Place, 6 Falcon Way, Shire Park
Welwyn Garden City,
AL7 1TW
United Kingdom of Great Britain and Northern Ireland
Packaging details and storage conditions
Packaging Type
Vial, Type I glass
Configuration
1x1
Shelf life (months)
36
Storage conditions
Do not store above 25°C, protect from light
API Manufacturing Site(s)
By Organization
By Active Ingredient
DSM Nutritional Products Ltd.
Zweigniederlassung Werk Sisseln, Hauptstrasse 4
Sisseln,
4334
Switzerland
Ceftriaxone
FPP Manufacturing Site(s)
F Hoffmann - La Roche Ltd
Wurmisweg
Kaiseraugst,
CH-4303
Switzerland